Latisse for Longer, Lusher Eyelashes
Latisse is an FDA-approved prescription treatment designed to produce fuller, thicker, and longer-looking eyelashes. Latisse – which is only available by prescription- is an excellent option for patients who may be frustrated or self-conscious about the appearance of short and/or sparse eyelashes. With regular applications over the course of 8 to 12 weeks, clients can expect to see longer and darker eyelashes that are ready to flutter and flirt!
Am I a Good Candidate For Latisse?
You’re a good candidate for Latisse if you have short, sparse, and/or light eyelashes. All candidates should be healthy individuals with no eye allergies, glaucoma, or any other eye conditions that may prevent them from using Latisse.
How Long Does a Typical Treatment Take?
Latisse is an incredibly convenient treatment, as every application can be done right in the comfort and security of your own home. It takes just a minute or two to apply the solution, which is placed right on the lash line. It’s recommended that you apply Latisse at night right before going to bed.

Consultations Are Available Now
Take the first step. Get your questions answered and find out more about Latisse by meeting with one of our knowledgeable medical staff at Boca Raton Plastic Surgery Center – Dr. Cristina Keusch for an in-person consultation.
Simply fill out the form on this page, and one of our experienced team members will reach out to you promptly to schedule your consultation. You can also call our Boca Raton, FL office directly to schedule (561) 368-9455.
Before And
After Gallery
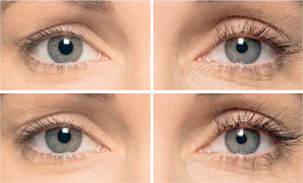
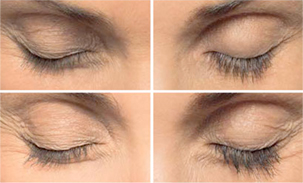

*Individual Results May Vary

What Results Will I See?*
Results can be pretty exciting, especially for clients who have felt frustrated or inconvenienced by their short eyelashes. Over the course of 8 to 12 weeks, clients should expect to see longer and darker-looking eyelashes that look and feel thicker. For many clients, that means kissing all that mascara goodbye – or applying it for even more of an “oomph”!
Is There Any Downtime?
There’s no downtime or recovery associated with Latisse; it’s a quick and easy topical solution that can be used right in your own home and incorporated into any nighttime routine.


